|
With 2-butanone a corresponding absorption occurs at 185 nm, which is out of the range of the spectrometer used to take the spectra. There is also an intense absorption band for 3-buten-2-one at 219 nm, which is a pi →pi ∗ transition. The absorption at 277 nm for 2-butanone is an n → pi ∗ transition, and with 3-buten-2-one, this absorption shifts to longer wavelengths (324 nm). 
The absorption frequency would be expected around 320 nm and 220 nm respectivley. La tabella degli assorbimenti IR è una carta sinottica da usare nell’ambito della spettroscopia infrarossa. For example, if you suspect that a compound is an eneone from its infrared spectrum, you easily could tell from the λ max of the n → pi ∗ and pi → pi ∗ absorption of the compound whether it is a conjugated eneone. A : The IR spectrum baseline may be curved due to the reasons below. 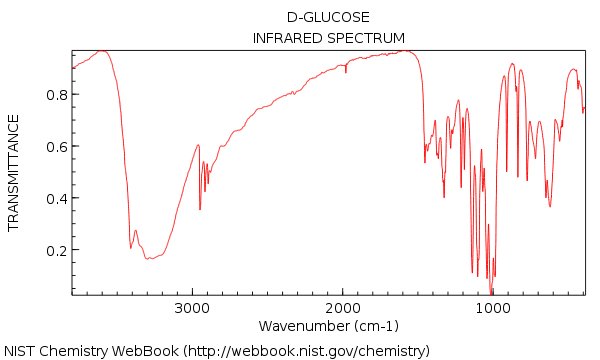
Neither of these absorptions is especially useful for specific identification unless the carbonyl group is conjugated, in which case the n → pi ∗ and pi → pi ∗ bands occur at longer wavelengths (by 30-40 nm). Microscope Transmission Spectrum of Magnesium Stearate. which corresponds to excitation of an electron from a pi-bonding orbital to a pi ∗-antibonding orbital. 
A more intense pi → pi∗ transition occurs about 180 -190 nm. Infrared spectra: Assignments of infrared frequencies are. \)Īldehydes and ketones absorb ultraviolet light in the region 275-295 nm, and the result is excitation of an unshared electron on oxygen to a higher energy level. H, C-O and CO were calculated and presented in Tables 1 to 3.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
